Automatic Full-Text Retrieval in Systematic Literature Reviews
Systematic Literature Reviews typically follow a 2-step process for screening literature. Step 1, where you identify relevant articles based on the title and abstract, and Step 2, where you investigate the full text of the identified articles for further analysis. While titles and abstracts are free to view, full texts papers are often not.
Key Pain Points
A lot of published research is open access and is freely available for public consumption. But this is not always the case, as some research may be published in traditional closed access (subscription-based) journals, and readers are charged a certain fee for access to the articles.
To conduct a robust, unbiased literature review, one must consider all relevant sources of data, open access or not. As a medical writer conducting literature reviews for a client, you may send the list of paid articles to your client, or if your organization conducts literature reviews in-house, you may have a separate team or process to procure these paid articles.
If you’re an experienced reviewer, you’d be familiar with the manual work and logistics behind a systematic literature review. This blog will focus on the logistics of identifying free vs paid articles and the associated archiving, post the title and abstract screening. Below is an example of a typical workflow:
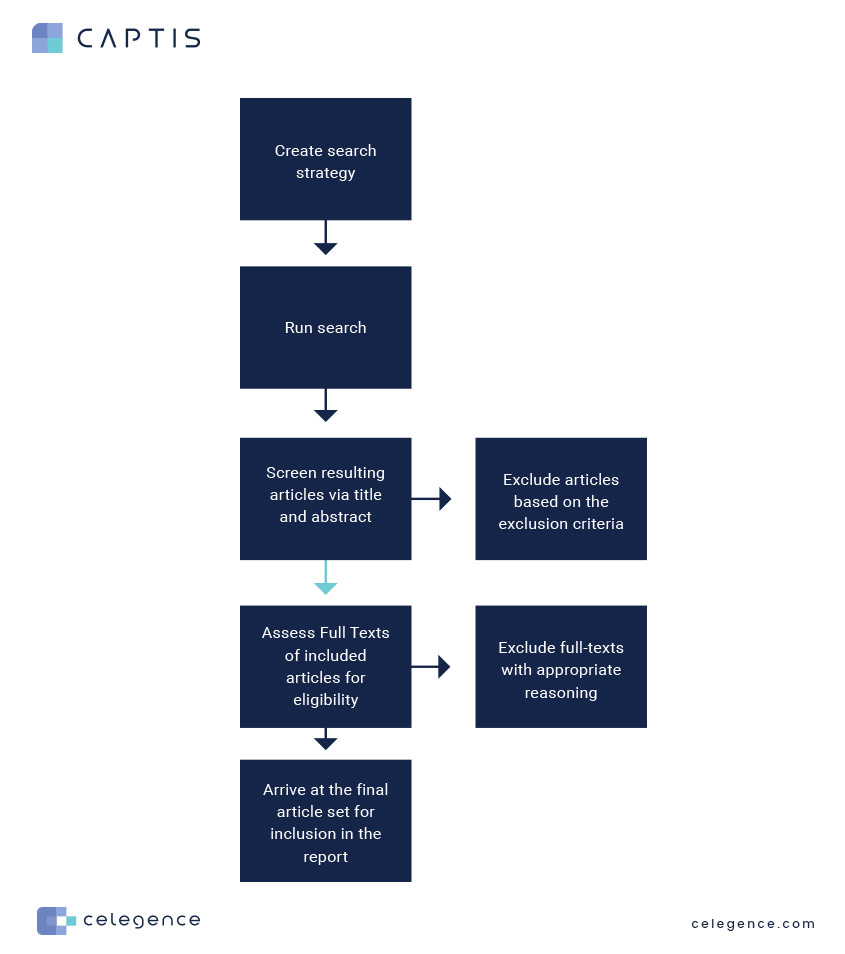
Now, let’s focus on the light blue arrow in the process flow above and estimate the manual effort behind progressing to full-text evaluation post the title and abstract screening stage.
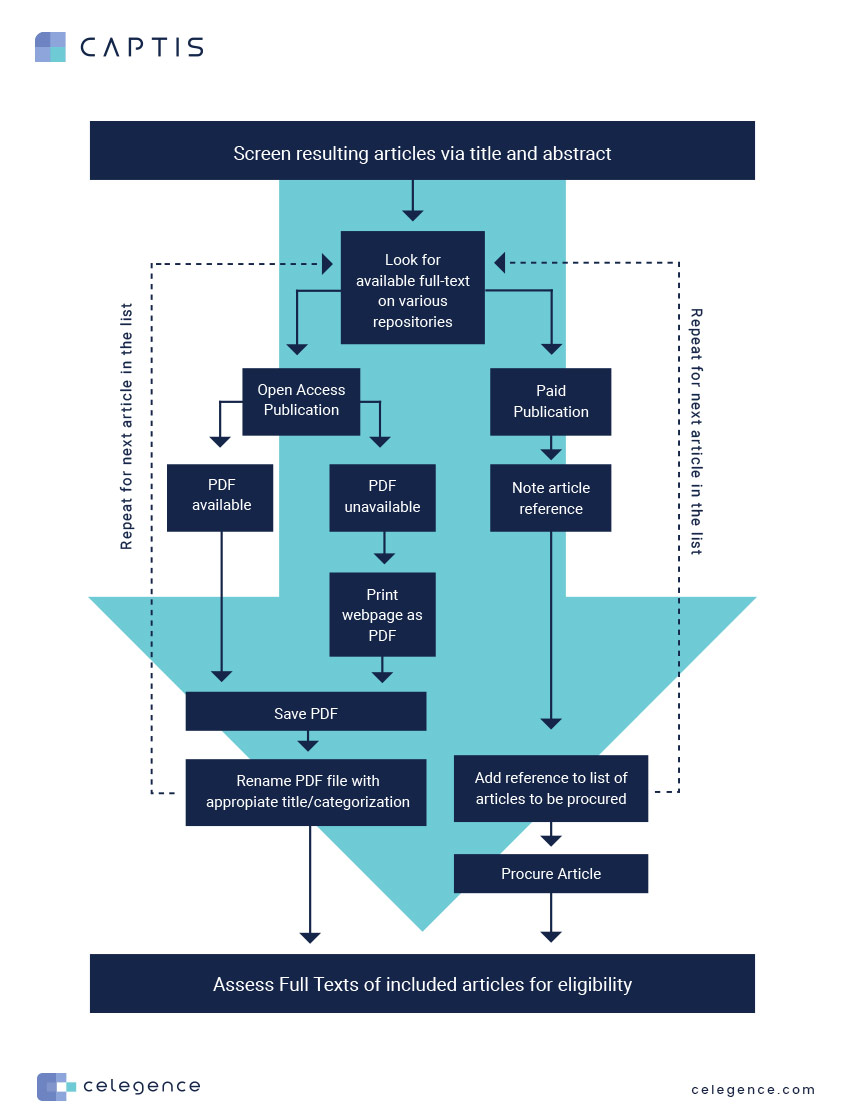
As evident from the flow chart above, gathering and compiling full-text references for the full-text evaluation is a multi-step, time-consuming process.
While many organizations have an internal repository of articles or subscriptions to paid journals, literature reviews for regulatory submissions such as Clinical Evaluation Report (CER) or Performance Evaluation Report (PER), must be submitted with full texts of all sources referenced in the document. Hence, the task of compiling a report-specific folder/location with PDFs of included literature references still remains.
The checklist highlights all of the documentation that you will need in place for certification of your IVD device and will serve as a guide to help you achieve ongoing compliance. In conjunction with this checklist, we are also able to provide you with bespoke strategies to bring your business up to speed. We are currently working with businesses from the United States, India, and throughout Europe to ensure that they are ready for the deadline in May of 2022.
Automatic Full-text Retrieval Within CAPTIS™
CAPTIS™ helps cut short the overall time for a literature review in many ways. Apart from automatically pulling in article metadata in your review dataset from sources like PubMed and Google Scholar, it also retrieves, renames, and stores any freely available full-text once the article is included in the title and abstract screening step.
In short, the tiresome process of searching for, downloading and storing full-text documents can now be eliminated.
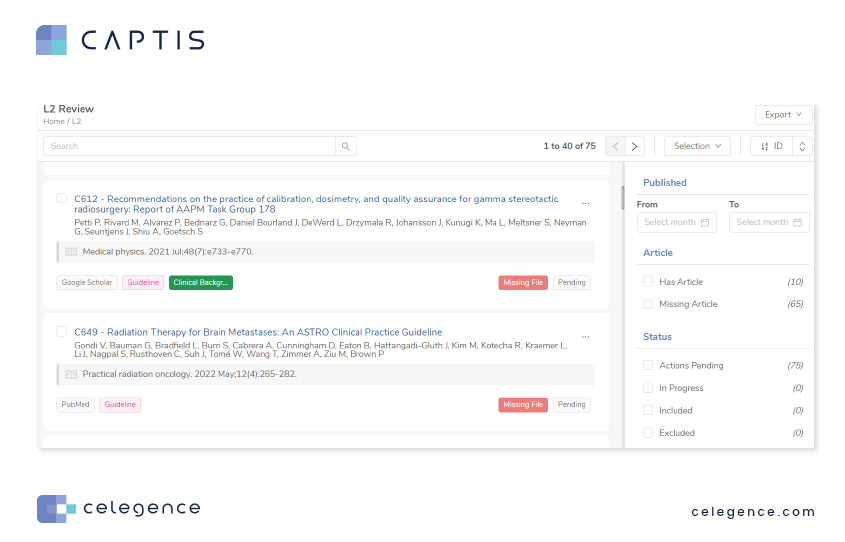
Using a unique built-in filter for article availability, CAPTIS™ automatically assigns tags to all records with missing files and allows you to filter them out using the Missing Article filter. This filtered list can then be exported out as an excel and shared with the concerned team for article procurement.
And once those missing PDFs are available, you can simply upload them back to CAPTIS™ on to their respective records. These PDFs will now be available for your current full-text review stage and any future reviews/cloned projects utilizing the same project.
Benefits of Automatic Full-text Retrieval within CAPTIS™
Here are some of the ways medical writers can save time using the automatic full-text retrieval feature within our EU & IVDR compliance tool CAPTIS™:
- Automatic identification and tagging of free vs paid articles: Writers need not check if full texts are freely available or not. CAPTIS™ does the background work for you and provides filters such as Missing Article to highlight which articles are missing PDFs
- Automatic download and renaming of available articles: CAPTIS™ automatically downloads, renames and attaches PDFs to relevant articles. This negates the logistical challenge of keeping articles/PDFs in the correct location, and in a singular format.
- Uploading articles: Manual uploads are automatically renamed by their CAPTIS™ article title, keeping the naming convention uniform throughout the project.
- Storage and download: All PDFs are attached to their respective record on CAPTIS™ and can be downloaded in a zipped folder when needed.
Schedule Your CAPTIS™ Demo
Your medical writing team can benefit from CAPTIS™ with faster turnaround times for systematic literature reviews and more accurate end-to-end MDR/IVDR documentation support. To learn more and view a comprehensive demo of CAPTIS™, reach out to info@celegence.com today or contact us online to connect with a Celegence representative.
The checklist highlights all of the documentation that you will need in place for certification of your IVD device and will serve as a guide to help you achieve ongoing compliance. In conjunction with this checklist, we are also able to provide you with bespoke strategies to bring your business up to speed. We are currently working with businesses from the United States, India, and throughout Europe to ensure that they are ready for the deadline in May of 2022.



