
Data Extraction Tables in CAPTIS™
Published scientific literature is an essential dataset reviewed as a part of the Clinical Evaluation for a medical device (Article 16 of the Medical Device Regulation (EU) 2017/745) or Performance Evaluation for an in-vitro diagnostic medical device (Annex XIII of In Vitro Diagnostic Medical Device Regulation (IVDR) (EU) 2017/746).
Once literature searches are executed, the adequacy of the searches should be verified and a literature search report should be compiled to present details of the execution, any deviations from the literature search protocol, and the results of the search. These data are best summarized in a data extraction table.
Literature reviews must be performed in a systematic manner. These reviews often involve a data extraction step wherein relevant pieces of information from the assessed studies are extracted in an organized manner to help analyze data and draw conclusions. Typical study parameters extracted include title, author, year, journal, aim, disease condition, patient population, follow-up duration, device used, performance outcomes, safety outcomes, study conclusion etc.
Presenting the extracted data in a tabular format allows for easy identification of key outcomes for reviewers and Notified Bodies alike. The tabular format also helps prepare for subsequent statistical analysis, if required. The data extraction table can either be attached as an appendix to the main document or be included as a part of the main literature section. This must be followed by a summary of the observed outcomes and overall conclusions from the dataset reviewed. Data extraction, while necessary, does require significant time and the process is prone to errors when performed manually.
The checklist highlights all of the documentation that you will need in place for certification of your IVD device and will serve as a guide to help you achieve ongoing compliance. In conjunction with this checklist, we are also able to provide you with bespoke strategies to bring your business up to speed. We are currently working with businesses from the United States, India, and throughout Europe to ensure that they are ready for the deadline in May of 2022.
Key Pain Points with Data Extraction Tables
Medical writers may need to keep multiple tools such as PDF files, Microsoft word files, Excel sheets with extracted article metadata open separately to extract data from the article. Manual data extraction features in our 101 Ways to Waste Time in a Systematic Literature Review. Frequently switching between multiple windows with manual checks such as ensuring that data is being extracted for the correct article can make this a time-consuming process. Moreover, this step is often preceded by another manual step where article metadata is extracted using an external software (reference management software are popular choices) or done manually (points casually at junior associates).
Pssst! Did you know CAPTIS can help you extract PubMed, Google Scholar, Europe PMC, Embase, Cochrane and Prospero article metadata with just a few clicks? Chat with us to learn how.
Data Extraction Tables Within CAPTIS
At Celegence, we understand operational challenges faced by device manufacturers and medical writers alike and use technology to overcome these. CAPTIS helps cut short the overall time for a systematic literature review in many ways.
CAPTIS allows you to easily configure custom data extraction tables for your literature review. You can choose between simple text boxes or configure a dropdown list of options to choose from when extracting data. Additionally, you can set up “Help Messages” for yourself or your team to have guidance handy whenever needed.
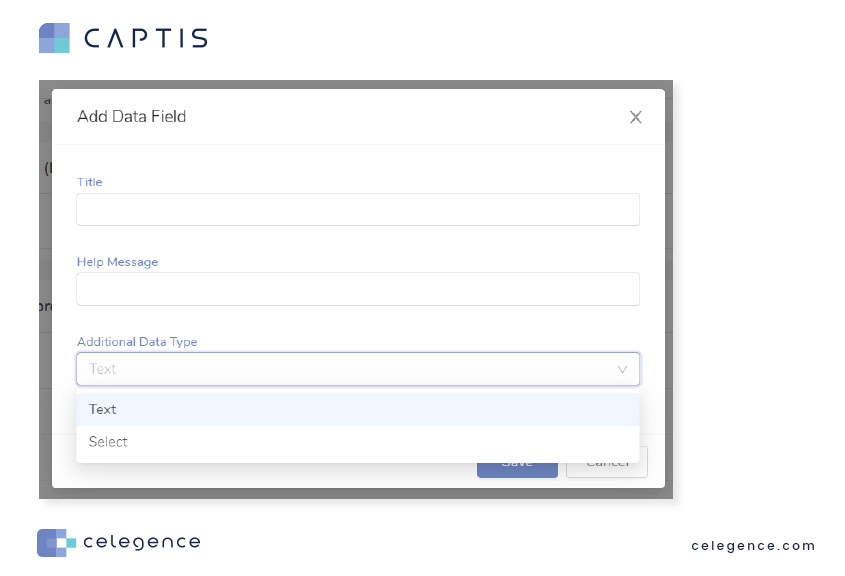
Data fields configured in the project settings can be saved as a template for reuse and appear on the right-hand side option panel when reviewing articles.
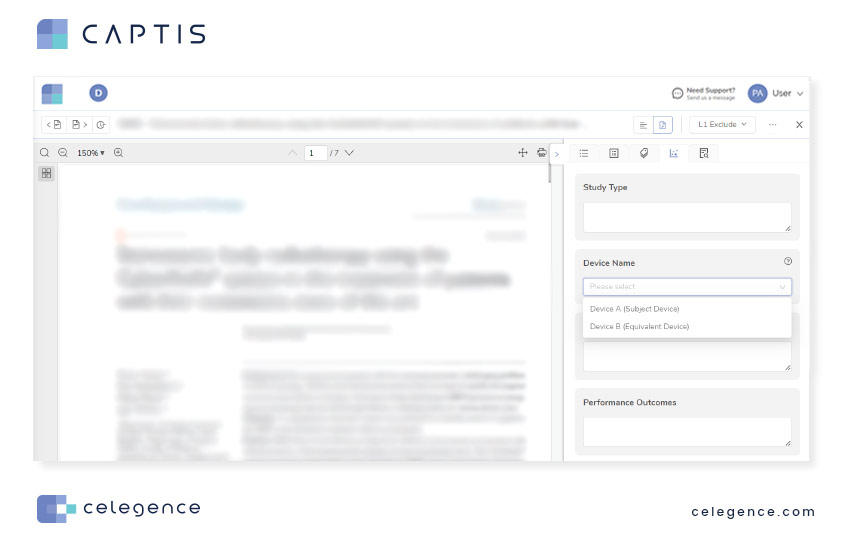
Data extraction on CAPTIS is faster than the manual method since bibliometrics information (i.e., publication types, journal names, authors, year of publications) of your entire project library is already stored in CAPTIS. The settings menu allows you to select which information to present for each article in your final data extraction table. The extracted data can be downloaded in the form of a tabular report for further usage.
Benefits of of Data Extraction Tables within CAPTIS
Here are some of the ways medical writers can save time using the data extraction table feature within CAPTIS:
- Customized data extraction – Users can set which data fields they want to collect for each article
- Set up templates on CAPTIS – All team members follow the same format for data extraction, maintaining consistency and quality.
- Summarize as your review – Data extraction options are right next to the article when performing literature reviews, allowing you to capture data as you review
- Single window approach – This web-based application displays all that you require for your literature review and extraction on the same platform, eliminating the need to open and switch between multiple application windows
- CAPTIS as a repository – The entire literature review and associated extracted data will always be on CAPTIS. This provides a centralized place to access your literature reviews, replacing excel sheets, documents, and system folders. Review data can be exported on demand in multiple formats, making CAPTIS an excellent archival tool.
Schedule Your CAPTIS Demo
Your medical writing team can benefit from CAPTIS with faster turnaround times for systematic literature reviews and more accurate end-to-end MDR/IVDR documentation support. To learn more and view a comprehensive demo of CAPTIS, reach out to info@celegence.com today or contact us online to connect with a Celegence representative.
The checklist highlights all of the documentation that you will need in place for certification of your IVD device and will serve as a guide to help you achieve ongoing compliance. In conjunction with this checklist, we are also able to provide you with bespoke strategies to bring your business up to speed. We are currently working with businesses from the United States, India, and throughout Europe to ensure that they are ready for the deadline in May of 2022.



